Exposing cells to controlled oxidative stress is believed to be a potentially powerful but as yet untapped method of inducing apoptosis (cell death), which can be applied to a range of scenarios including cancer treatment or anti-microbial function. The challenge is create oxidative stress in a controlled way, whereby three factors are controlled simultaneously: the location, the timing, and the quantity of the produced Reactive Oxygen Species (ROS). The aim of this work was to demonstrate the ability to produce ROS by a controlled enzymatic reaction, using liposome-based microcapsules whose ability to selectively bind to the lablet surface has been demonstrated previously (cf. D9.1 Report), loaded with a precursor of the ROS, and coupled with enzyme immobilised in cross-linked gel particles that can also reside at the lablet surface and which catalyse the single-electron oxidation of the substrate to produce ROS. Due to high reactivity of the ROS, the sequence of events – release of substrate, enzymatic oxidation, attack of a cell wall – must occur in close proximity, and is therefore suitable to mobile, independent agents such as the lablets.
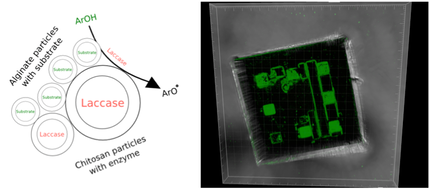
Figure 1: (a) Concept of in-situ formation of Reactive Oxygen Species by enzymatic reaction from stable precursors released from microcapsules that can adhere to the lablet surface. (b) Lablet with electrodes occupied by locally concentrated capsules able to carry ABTS substrate. The capsules are labelled by green fluorescein.
The challenge in making this concept work is finding the right combination of parameters that ensure functionality of all sub-systems (cf. Fig. 1). Variables such as the type and quantity of the substrate, the concentration of the enzyme, diffusion properties of the microcapsules, and the environmental variables (e.g. temperature) represent a parametrically very rich problem. The aim of the work was to explore this vast number of parameter combinations and to optimise the enzyme coating composition and reaction conditions so as to achieve a specific function – the ROS attack of cancer cells.